Kathy Fay Mahdoubi
Nov 05, 2013
The field of molecular imaging is grazed by two different animals—the workhorses, such as SPECT, PET and tried-and-true biomarkers like FDG, and the young, intrepid and unpredictable newcomers that have been given a name, taken around the track a couple times, but perhaps are still too fresh to be saddled with the responsibility of routine clinical use. These are the innovations in dementia and psychiatric imaging, hybrid and optical-acoustic modalities and anatomy-specific precision biomarkers. While the herd of the latter is extensive, only a few are notable enough to include here amongst the most promising workhorses five or more years from now. What technologies and biomarkers are going to make it through the gate to race into clinical practice?
Molecular Imaging Insight caught up with four members of the World Molecular Imaging Society’s (WMIS) leadership, all experts from top institutions across the U.S., during the recent World Imaging Society Congress (Sept. 18-21) in Savannah, Ga. After reviewing the year’s abstracts and presentations for the meeting, each had perspectives to offer about what’s in store over the next several years.
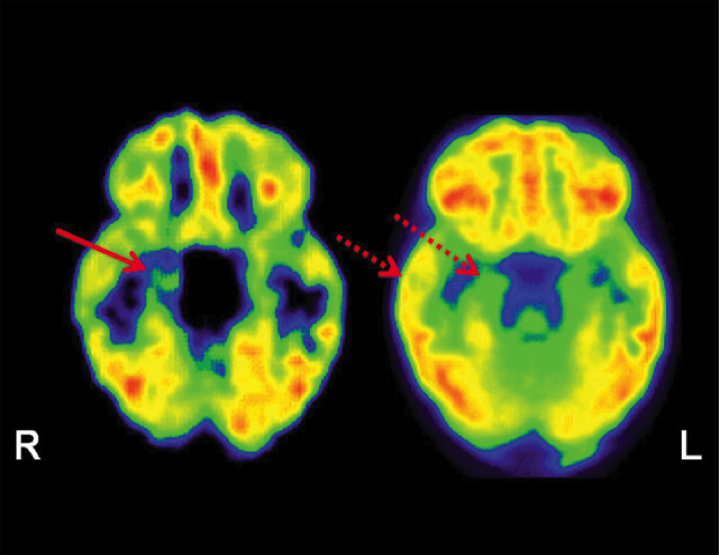
C-11 flumazenil PET (left image) showing focal abnormality (decreased tracer binding) involving right hippocampus (solid arrow) in patient with medial temporal lobe epilepsy. In comparison, F-18 FDG PET (right image) shows widespread hypometabolism in right temporal lobe, including neocortex (broken arrows).
— Journal of Nuclear Medicine
Among the most novel and watched radiopharmaceuticals today are F-18 FLT, NAV4694, I-123 MIBG and Ga-68 DOTATOC. But a few more are gaining believers as well. The amino-acid imaging agent F-18 FDCBC and synthetic anti-3-F-18-FACBC are very active in prostate cancer studies and recently shown to best C-11 choline for imaging radically treated prostate cancer (Eur J Nucl Med Mol Imaging July 2013). Another notable is F-18 Fluorodopa. This molecular imaging probe is no stranger to the research community, recently proving its value in imaging gliomas (J Neurooncol 2013 Jan;111(1):11-8). F-18 Fluorodopa has additionally been involved in research of movement disorders including Parkinson’s (Parkinsonism Relat Disord 2012 Jan;18 Suppl 1:S96-9). These are also ones to watch in the realm of translational research.
As of this year, both U.S. and European regulatory bodies have approved Radium-223 dichloride (Xofigo) to treat metastatic castration-resistance prostate cancer. The clinical use of this therapeutic drug is set to expand and is already being used in investigative cases of metastatic breast tumors. According to a September 2013 report from BioTrends Research Group, almost 25 percent of surveyed oncologists in the U.S. are prescribing the drug and nearly half of those not current prescribing expect to be within the next six months.
Another concept presented as an area gaining strength is companion diagnostics, a sophisticated term for the marriage of therapeutic and diagnostic agents. While this is not particularly new, the emergence of increasingly complex and radical treatments and rapid treatment monitoring could gain considerable strength in the years to come.
Optical imaging
Moving from biomarkers to optical imaging, we see nano-carriers, such as those employing indocyanine green dye (ICG) as an agent for near-infrared fluorescence optical imaging, gaining attention in the context of diagnostic imaging and therapy monitoring, and drug screening, due to its depth of imaging and low absorption (J Nanomed Nanotech 2012, 3:8).
Optical imaging has not made it into the average surgical center, but that is not a concern for Anna M. Wu, PhD, current WMIS president, professor and vice chair of molecular and medical pharmacology at the David Geffen School of Medicine at UCLA, Los Angeles. Wu says optical research has a strong presence in the newest literature for applications in seeking out malignancies. The optical imaging technologies currently being used in clinical practice are just a scratch on the surface for this discipline. The WMIS as a collective is aiming higher.
“More general fluorescent probes like ICG are FDA approved and being used for certain applications. But what we are pushing are probes that are very molecularly specific so that the technique being used is the same, despite the quality of information you get being at a completely different level,” explains Wu.
An example would be agents that home in on positive and negative controls for HER1, HER2 and beta-D-galactose receptors, providing a means of not only screening for malignancies but mining concurrent information about those tumors (Methods Mol Biol 2012;872:191-204).
Surgery is beginning to explore optical imaging and watching development of novel optical imaging agents. “There has been continued growth in chemical agents for optical imaging,” says Mark D. “Marty” Pagel, PhD, program chair for the 2014 World Molecular Imaging Congress (WMIC) in Seoul, Korea, and a professor of biomedical engineering, chemistry and medical imaging at the University of Arizona in Phoenix, Ariz. He notes that while the body produces chemicals that interact with light, it’s more informative to use a material that can be injected into an animal model or a human patient to interact with novel enzymes and metabolites involved in cancer and diabetes.
“The interest in intra-surgical imaging has really taken off. We have an interest group in this area,” says Pagel. “By combining these chemical probes, you can now paint the tissue as it is being exposed during surgery and surgeons put on their fluorescent goggles and say, ‘Oh, I didn’t cut out the whole tumor. I have to go back and take more.’ I think it is a brilliant idea, no pun intended.”
So popular was that intra-surgical interest group at the 2013 WMIC that as many as 100 people attended the discussion. This is an area that shows great promise for further clinical adoption.
Move over conventional MRI
Another area developing new instrumentation is magnetic resonance, but not just any MR—a super-charged molecular version of MR that is entirely molecular and even atomic, rather than looking at water or fatty tissues with conventional MR.
“The way to get around that is to do a hyperpolarization technique that involves a lot of physics,” Pagel says. “They cool the material down nearly to absolute zero and then very rapidly warm it up and make sure it’s at the right mediation and inject it into the body. It’s quite a technological feat and a lot of engineers have put a lot of time into this. By hyperpolarizing, you get a huge amount of signal from one particular carbon atom in a molecule and all the other carbon atoms are effectively silent and cannot be detected. It’s great for tracking that one hyperpolarized carbon as it stacks from one molecule to another.”
One such application for this is targeting hyperpolarized vitamin D to monitor oxidants in the body, but in this space, metabolic imaging may just trump all. “This area of MR research—hyperpolarized MR—involves probes that can really look at real-time metabolic imaging of disease and their translation to the heart and diabetes, to cancer and the brain,” says Jason S. Lewis, PhD, program chair of the 2013 WMIC, chief of radiochemistry and imaging science service and vice chair of radiology research at Memorial Sloan Kettering Cancer Center in New York City.
“Its biggest application has been in imaging the glucose consumption in tumors—glucose and lactate,” remarks Pagel, who recounted a WMIC session discussing how hyperpolarized carbon atoms were followed with MR for the localization of benign and malignant prostate cancer. As you might imagine, this concept has a lot of potential for other kinds of metabolic applications, too, most notably diabetes imaging. “Being a metabolic disease, diabetes is ideal for molecular imaging—we have to detect those metabolites,” says Pagel. “There is a good role for MRI here, because diabetes involves organs that are soft in tissue composition. MRI is really good at looking at big juicy organs. By tracking the general metabolism or specific metabolites with diabetes that can be very helpful. There are ions involved in diabetes and tracking the flow of ions into and out of certain areas of the body also is helpful for diabetes research.”
All three experts agree that the possibilities of molecular MRI are expansive. “This is emerging and a lot of centers are doing preclinical research around the world and also now getting into it clinically, so this is something we will for sure see a lot more of next year,” notes Lewis.
Methods of interest
We cannot talk about the future of molecular imaging without talking about PET/MR. Experts agree it could take several years to carve out a permanent space for this attractive duo. It is still uncertain whether PET/MR will have the same impact that PET/CT has had in terms of widespread and relatively rapid adoption, but there are several indications that the hybrid will have an impact in other areas including oncologic and pediatric imaging (Radiology April 2013 267:1 26-44).
“PET/MR is just a phenomenal tool—it is the combination of two very strong technologies,” says Lewis. “Both have limitations and advantages and they provide very powerful scientific data.”
Group consensus favors the advancement of PET/MR in neuroimaging, and specifically for functional imaging of brain circuitry. “A number of PET imaging methods can be shown to look at the molecular detail in the brain and they are then marrying that with MRI to look at brain function,” adds Pagel.
The clinical indication of choice seems to be in oncologic applications, especially head and neck cancer. PET/MR also is gaining traction in areas of cardiac imaging and non-oncologic neuroimaging, according to a report in the September issue of Current Radiology Reports.
Since it came on the scene for clinical neuroimaging in 2007, PET/MR has slowly reached the position of being considered a viable hybrid imaging system for clinical use and not simply a powerful tool for research. The review noted an unpublished independent multireader study comparing FDG PET/CT and PET/MR in the stages of head and neck neoplasms. In the study, PET/MR correctly identified 80 percent of the FDG-avid lesions caught by PET/CT and there was complete inter-reader agreement in staging documented for the 11 out of the 14 subjects in the study. PET/MR found additional lymph node involvement in the discrepant cases.
Neuroimaging round-up
A wealth of other neurological research was revealed this year and with it, several possibilities for molecular brain imaging. “There is a range of neuro-applications, from drug addiction to ischemia and loss of blood pressure in the brain,” lists Pagel. “Alzheimer’s disease is a big topic, of course, and now linking the molecular causes of psychiatric diseases and that’s a huge issue.”
One of this year’s groundbreaking studies shows promise in adenosine SPECT not only for neurodegenerative disorders such as Parkinson’s and Huntington’s disease, but for psychiatric imaging of schizophrenia, bipolar disorder, chronic depression, anxiety and other mood disorders with the help of I-123 MNI-420. In conjunction with SPECT, this agent seeks out A2A receptors involved in dopaminergic systems in the brain that are associated with a range of neurological disorders (J Nucl Med 2013; 54:1–8).
Amyloid imaging was both expanded and took a hit, depending on your perspective, following the final CMS decision to allow coverage of one amyloid PET scan per patient in government appointed clinical trials (coverage with evidence development, or CED).
As you’ll see on page 10, SNMMI President-elect Peter Herscovitch talks about the implications of this decision and how the industry is bound to engage in novel neuroimaging research, including the creation of extensive trials and potential registries similar to the National Oncologic PET Registry (NOPR) for oncologic PET under CED.
NOPR was supported by the WMIS in conjunction with the American College of Radiology (ACR) and the American College of Radiology Imaging Network (ACRIN). Lisa Baird, WMIS executive director, says her organization plans to carry that forward for dementia imaging.
“We sponsored oncology trials through NOPR and we look forward to doing the same for amyloid [imaging], as well, to organize the coverage with evidence development studies that are needed,” says Baird. “Even once the imaging agents are approved by the FDA, in order for them to be widely and effectively used, there needs to be reimbursement and acceptance by CMS and insurers that using these targeted scans is of value in terms of identifying and managing disease. The WMIS takes a very active role alongside ACR and ACRIN to put together registries for CMS.”
Lewis asserts that this really shouldn’t be the primary focus of researchers and radiopharmaceutical companies that could get bogged down by regulatory drag. “That’s almost at the end point, but at the beginning point—to be seeing the cutting edge technologies, the new probes, the new devices and everything else, to follow that through really helps investigators.”
Challenge of gaining and retaining funding
On the minds of WMIS leadership was a nagging concern for the lack of funding for new and critical research. As of March 2013, 5 percent of National Institutes of Health (NIH) funding was contracted, or sequestered, according to presidential mandate. That means $1.55 billion of its fiscal year budget. Since the NIH is the single largest funder of biomedical research the world over, this pinch is being felt not only by molecular imaging specialists, but scientists everywhere.
“Lack of NIH funding is a major challenge, as it is a critical resource for new research,” comments Lewis. “We have some exceptionally brilliant young minds and [NIH funding] really helps them find their path and build up innovative and exciting research programs. We help them tap into all the resources available, but I do think that is a challenge.”
It is not just about exhibiting all that is state-of-the-art on the research bench; it’s about expounding upon and translating these applications. Intra-surgical optical imaging will no doubt continue to develop, as will clinical use of PET/MR and hyperpolarization MR techniques. Companion diagnostics will most likely gain increasing importance as new applications of predictive and perioperative imaging mature, for instance in cases of radioimmunotherapy and radioembolization, and new biomarkers for hypoxia and amino acid imaging look to be on the horizon for regulatory approval.
“I think that we as a field realize that the ultimate goal is the get these approaches and these molecular imaging technologies into clinical use, but there are many steps along the way,” Wu concludes.
This article has been posted with the permission of Molecular Imaging Insights.
Article link: http://www.molecularimaging.net/topics/molecular-imaging/biomarkers/perspectives-promising-research-whats-next?nopaging=1
